Silhouet Tone RMD Structural Remodeling System 411570
FACELIFT WITHOUT SURGERY
Structural Remodelling System
The RMD is a unique rejuvenation system that aims to improve one’s well-being by significantly reducing the signs of aging through non-invasive, non-surgical means. Our device is designed to specifically support the treatment of skin texture, smoothing age-related expression lines, tightening pores, and toning facial and neck muscles.
The RMD is proven to firm sagging muscles, increase local blood and lymph circulation, stimulate fibroblast activity, increase the synthesis of new collagen, repair existing collagen and restore the homeostasis of skin cells.
Five technologies | One device
Main features of the RMD
- Anti-aging system of its kind.
- Combinations of exclusive integrated technologies for skin and muscle firming.
- 5 exclusive technologies in one device.
- Effects on the dermis, epidermis and muscles.
- Painless, non-invasive treatment.
- Independent of your skin colour.
- Achievable throughout the year.
- It does not require prior skin preparation and is performed without the need for anesthesia.
- Does not require post-treatment cooling.
- Ergonomic handpiece.
- Easy to use, modern design.
- Designed and manufactured in North America with a high-quality after-sales service.
Main advantages
- Health Canada cleared a Class II Medical Device.
- Clinically-proven, visible results achieved within 2 to 8 treatments.
- Non-invasive, safe, effective and efficient.
- Designed and manufactured in Canada in compliance with the highest medical standards.
- Proprietary programmed integration of technologies.
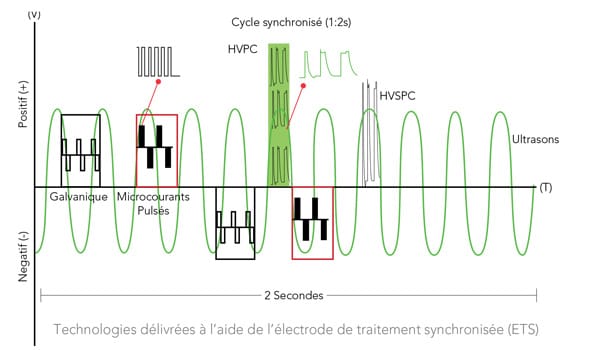
The technologies behind the structural remodelling system
The RMD: Structural Remodelling System is designed to deliver the most remarkable, non-invasive skin remodelling results in the world of medical esthetics. This device makes optimal, safe, and effective use of 5 recognized electrotherapy technologies, exclusively designed to work in synergy and deliver outstanding results.
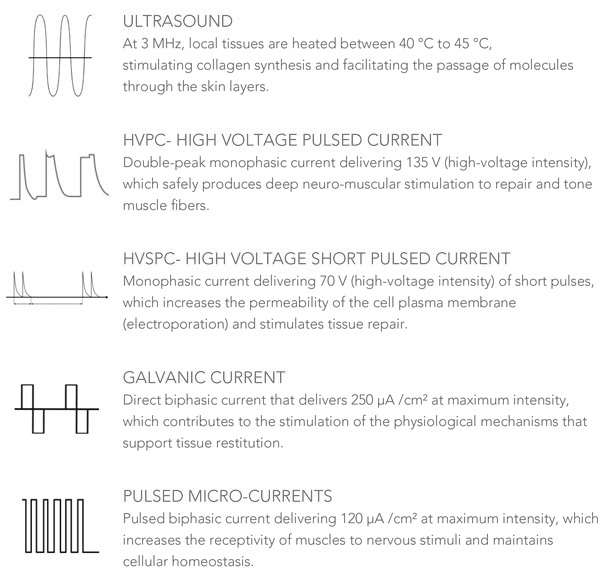
Synchronized Treatment Head (STH)
The RMD operates with an exclusive Synchronized Treatment Head (STH) that effectively delivers energy to the dermis and muscle tissue, where it safely elicits responses that tone the muscle and rejuvenate the skin.
- Effective.
- Safe.
- In unique cyclical programming.
- Linked and electronically controlled by a program with adjustable parameters according to individual tolerance level.

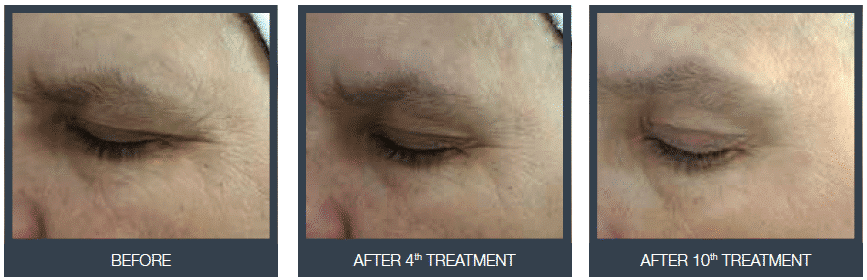
Treatment in action
Our trainer practices a structural remodelling treatment at the facial level.
Outstanding results and immediate effectiveness
Improvement of skin structure
Skin structures densification.
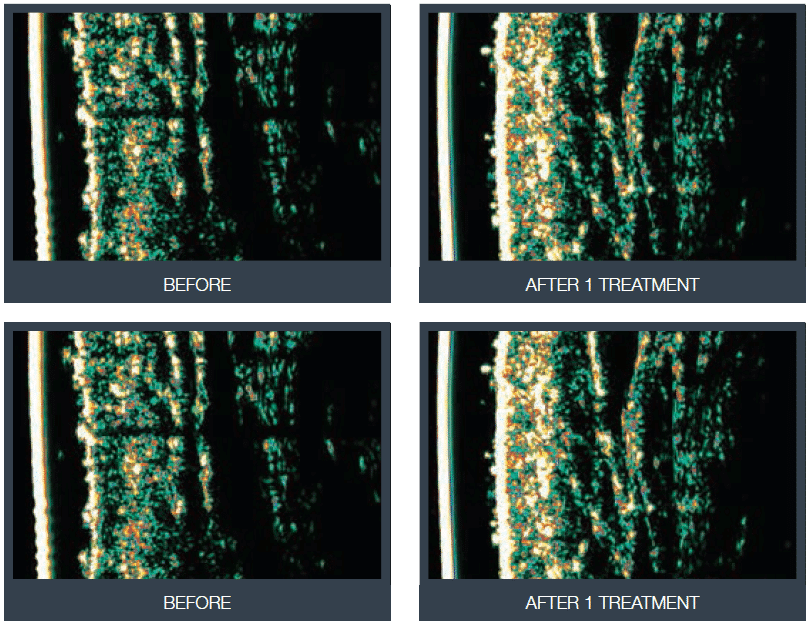
Pore improvement
Pore improvement exceeded an average of 119% for the case-group participants with an average age improvement of 3 years and, in some cases, of up to 6 years.
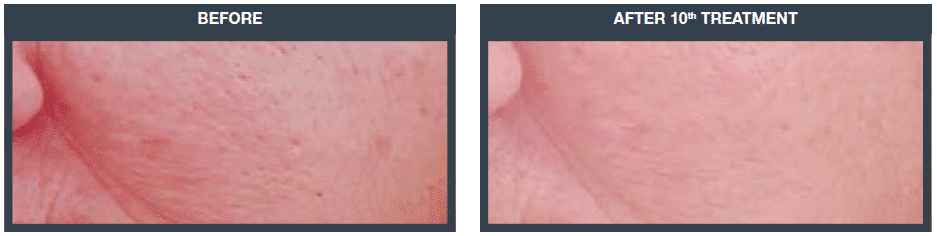
Wrinkle reduction (frown lines)
Wrinkle reduction improved, on average, by 151% for group participants, with an average age improvement of over 5 years, and in some cases, up to 9 years.
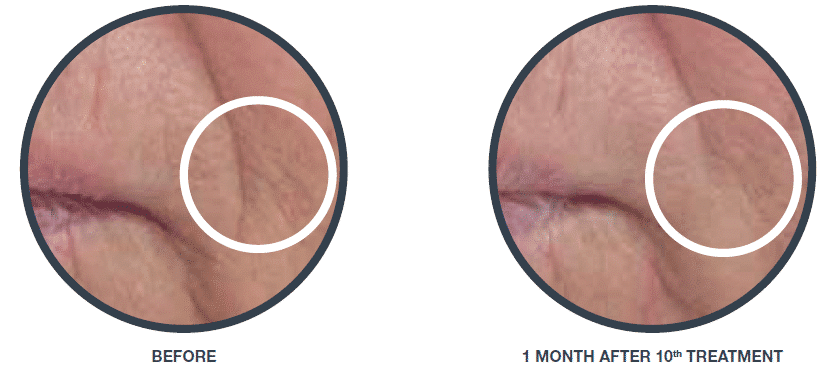
 Wrinkle reduction (nasolabial fold)
Wrinkle reduction (nasolabial fold)
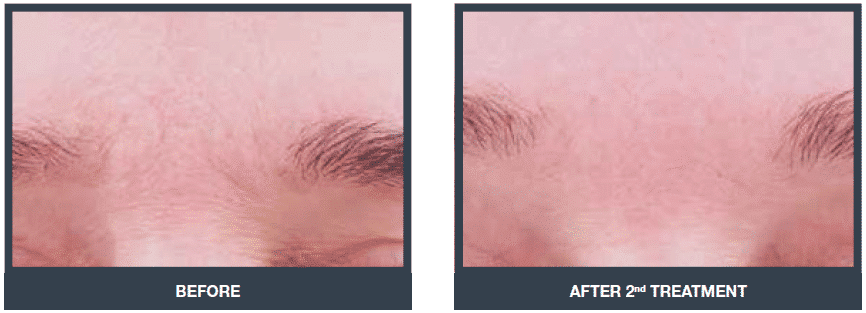
Muscle strengthening (eyebrow lifting)
- All photographs were taken with a Canfield Scientific VISIA imaging system and skin analysis.
RMD training
Our training will allow you to discover in-depth the medical and aesthetic care essential to your institute.
Here is our lesson plan:
- Advantages and benefits of the microdermabrasion device.
- Benefits of having five technologies all in one device.
- Theory.
- Technique and method of work.
- Treatment protocols in the institute.
- Sales and Marketing Introduction.
- Establish a treatment schedule with clients.
- Practice.
* Prerequisite: Diploma in aesthetics
* Duration: a full day
Why is the RMD essential to your institute?
Skin care professional benefits
- Good return on investment
- Safe
- Easy to use
Client benefits
- Instant and visible results
- Minimal downtime
- Painless
- Five integrated technologies.
- Acquiring new potential customers and boosting our existing clientele.
- Recognized effectiveness in the aesthetic and medical fields and with consumers.
The benefits of having the RMD in your institute
Shipping fees are Not Included in the price they will be calculated and added separately. / Les frais de livraisons ne sont pas inclus dans le prix de vente et seront rajouté séparement.
FACELIFT WITHOUT SURGERY
Structural Remodelling System
The RMD is a unique rejuvenation system that aims to improve one’s well-being by significantly reducing the signs of aging through non-invasive, non-surgical means. Our device is designed to specifically support the treatment of skin texture, smoothing age-related expression lines, tightening pores, and toning facial and neck muscles.
The RMD is proven to firm sagging muscles, increase local blood and lymph circulation, stimulate fibroblast activity, increase the synthesis of new collagen, repair existing collagen and restore the homeostasis of skin cells.
Five technologies | One device
Main features of the RMD
- Anti-aging system of its kind.
- Combinations of exclusive integrated technologies for skin and muscle firming.
- 5 exclusive technologies in one device.
- Effects on the dermis, epidermis and muscles.
- Painless, non-invasive treatment.
- Independent of your skin colour.
- Achievable throughout the year.
- It does not require prior skin preparation and is performed without the need for anesthesia.
- Does not require post-treatment cooling.
- Ergonomic handpiece.
- Easy to use, modern design.
- Designed and manufactured in North America with a high-quality after-sales service.
Main advantages
- Health Canada cleared a Class II Medical Device.
- Clinically-proven, visible results achieved within 2 to 8 treatments.
- Non-invasive, safe, effective and efficient.
- Designed and manufactured in Canada in compliance with the highest medical standards.
- Proprietary programmed integration of technologies.

The technologies behind the structural remodelling system
The RMD: Structural Remodelling System is designed to deliver the most remarkable, non-invasive skin remodelling results in the world of medical esthetics. This device makes optimal, safe, and effective use of 5 recognized electrotherapy technologies, exclusively designed to work in synergy and deliver outstanding results.

Synchronized Treatment Head (STH)
The RMD operates with an exclusive Synchronized Treatment Head (STH) that effectively delivers energy to the dermis and muscle tissue, where it safely elicits responses that tone the muscle and rejuvenate the skin.
- Effective.
- Safe.
- In unique cyclical programming.
- Linked and electronically controlled by a program with adjustable parameters according to individual tolerance level.

Treatment in action
Our trainer practices a structural remodelling treatment at the facial level.
Outstanding results and immediate effectiveness
Improvement of skin structure
Skin structures densification.

Pore improvement
Pore improvement exceeded an average of 119% for the case-group participants with an average age improvement of 3 years and, in some cases, of up to 6 years.

Wrinkle reduction (frown lines)
Wrinkle reduction improved, on average, by 151% for group participants, with an average age improvement of over 5 years, and in some cases, up to 9 years.
 Wrinkle reduction (nasolabial fold)
Wrinkle reduction (nasolabial fold)

Muscle strengthening (eyebrow lifting)

- All photographs were taken with a Canfield Scientific VISIA imaging system and skin analysis.
RMD training
Our training will allow you to discover in-depth the medical and aesthetic care essential to your institute.
Here is our lesson plan:
- Advantages and benefits of the microdermabrasion device.
- Benefits of having five technologies all in one device.
- Theory.
- Technique and method of work.
- Treatment protocols in the institute.
- Sales and Marketing Introduction.
- Establish a treatment schedule with clients.
- Practice.
* Prerequisite: Diploma in aesthetics
* Duration: a full day
Why is the RMD essential to your institute?
Skin care professional benefits
- Good return on investment
- Safe
- Easy to use
Client benefits
- Instant and visible results
- Minimal downtime
- Painless
- Five integrated technologies.
- Acquiring new potential customers and boosting our existing clientele.
- Recognized effectiveness in the aesthetic and medical fields and with consumers.




